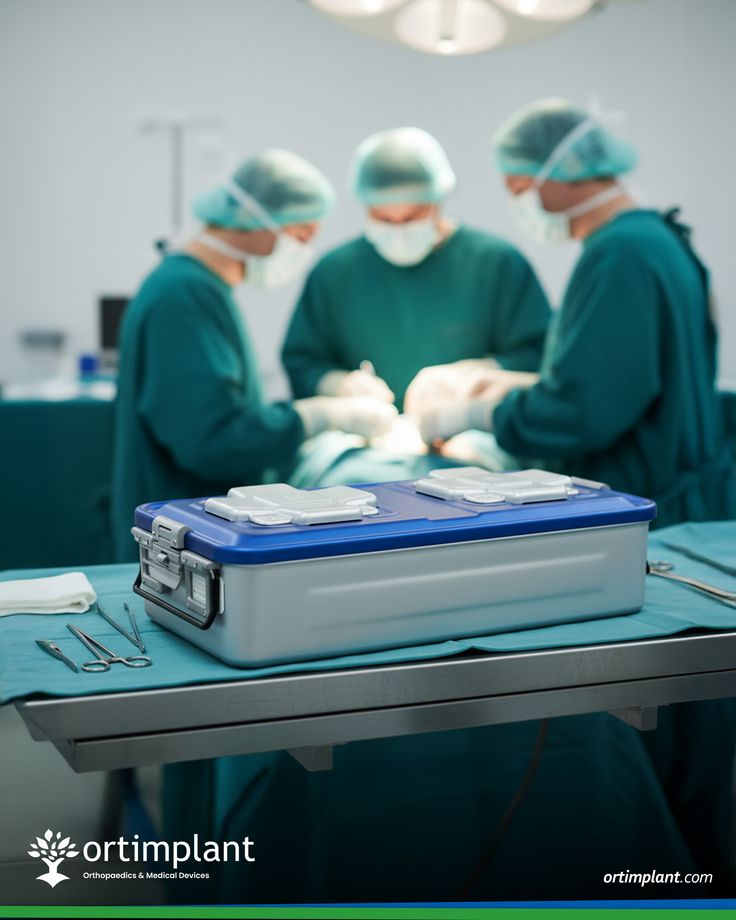
In Australia’s highly regulated healthcare landscape, ensuring safety and compliance is non-negotiable, and a sterile barrier system plays a central role in achieving that goal. Medical providers, manufacturers, and distributors rely on sterile barrier system solutions to protect devices from contamination while maintaining affordability. As healthcare costs rise, businesses are looking for sterile barrier system options that balance compliance with budget-conscious strategies. With growing awareness around infection control, the sterile barrier system has become a cornerstone of trust and reliability in the industry.
What is a Sterile Barrier System and Why It Matters
A sterile barrier system is designed to prevent microorganisms from entering and contaminating medical devices, making it essential for patient safety. In Australia, strict regulatory frameworks demand that every sterile barrier system meets high standards for durability and integrity. From surgical tools to diagnostic kits, a sterile barrier system ensures that products remain sterile until the point of use. Businesses that invest in a reliable sterile barrier system not only meet compliance requirements but also build credibility with healthcare providers seeking dependable solutions.
Key Components of Sterile Packaging Systems
A sterile barrier system is made up of several critical elements, including sterile packaging systems, sealing processes, and microbial barrier materials. These sterile packaging systems are engineered to maintain sterility under varying environmental conditions. Microbial barrier materials used in a sterile barrier system must resist tears, punctures, and moisture to ensure consistent performance. By integrating advanced sterile packaging systems, manufacturers can deliver a sterile barrier system that meets both safety and cost expectations.
Understanding Microbial Barrier Materials
Microbial barrier materials are the backbone of any sterile barrier system, providing protection against bacteria and other contaminants. In Australia, selecting the right microbial barrier materials for a sterile barrier system is crucial for compliance with healthcare standards. These materials are tested rigorously to ensure they maintain integrity throughout storage and transportation. A high-quality sterile barrier system uses advanced microbial barrier materials to deliver long-lasting protection while keeping costs manageable for businesses.
Medical Device Packaging Integrity Explained
Medical device packaging integrity is a critical factor in determining the effectiveness of a sterile barrier system. Without proper sealing and durability, a sterile barrier system can fail, leading to contamination risks. Australian healthcare providers prioritize sterile barrier system solutions that demonstrate proven medical device packaging integrity through testing and validation. Ensuring medical device packaging integrity within a sterile barrier system helps businesses avoid costly recalls and maintain trust with their customers.
Aseptic Barrier Packaging Solutions for Modern Needs
Aseptic barrier packaging solutions are evolving rapidly, offering improved efficiency and reliability within a sterile barrier system. These solutions focus on maintaining sterility without compromising usability or cost. In Australia, companies are adopting aseptic barrier packaging solutions to enhance their sterile barrier system performance while meeting regulatory requirements. By leveraging innovative aseptic barrier packaging solutions, businesses can optimize their sterile barrier system for both safety and affordability.
Contamination Prevention Packaging Technology Trends
Contamination prevention packaging technology is advancing, making the sterile barrier system more effective than ever. Innovations in contamination prevention packaging technology include improved sealing techniques and stronger materials that enhance sterile barrier system performance. Australian companies are increasingly investing in contamination prevention packaging technology to stay competitive and compliant. A modern sterile barrier system integrates these technologies to provide robust protection while minimizing operational costs.
Cost-Effective Sterile Barrier System Solutions
For price-conscious businesses in Australia, finding a cost-effective sterile barrier system is essential. Balancing quality and affordability requires careful selection of materials and processes. A well-designed sterile barrier system reduces waste, improves efficiency, and lowers long-term costs. By choosing the right sterile packaging systems and microbial barrier materials, companies can achieve a sterile barrier system that delivers value without compromising safety.
Building Trust Through Reliable Packaging
Trust is a major factor in the healthcare industry, and a dependable sterile barrier system plays a significant role in building that trust. Healthcare providers expect consistent performance from every sterile barrier system they use. By ensuring medical device packaging integrity and using high-quality contamination prevention packaging technology, businesses can strengthen their reputation. A reliable sterile barrier system not only protects patients but also reinforces confidence among stakeholders.
Why Australian Businesses Choose Advanced Solutions
Australian companies are increasingly turning to advanced sterile barrier system solutions to meet growing demands. These solutions incorporate cutting-edge aseptic barrier packaging solutions and contamination prevention packaging technology. A modern sterile barrier system allows businesses to stay compliant while optimizing costs. By investing in innovative sterile packaging systems, organizations can future-proof their sterile barrier system strategies.
The Role of vanderstahl in Sterile Barrier Innovation
The brand vanderstahl has emerged as a trusted name in delivering high-quality sterile barrier system solutions tailored for modern healthcare needs. By focusing on durable microbial barrier materials and efficient sterile packaging systems, vanderstahl helps businesses achieve reliable sterile barrier system performance. Their commitment to medical device packaging integrity ensures that every sterile barrier system meets stringent standards while remaining cost-effective. For Australian companies seeking trustworthy partners, vanderstahl provides solutions that align with both compliance and budget requirements.
Choosing the Right Sterile Barrier System Provider
Selecting the right provider for a sterile barrier system is crucial for long-term success. Businesses should evaluate expertise in sterile packaging systems, quality of microbial barrier materials, and proven track record in medical device packaging integrity. A strong sterile barrier system provider will also offer innovative aseptic barrier packaging solutions and advanced contamination prevention packaging technology. By making informed decisions, companies can ensure their sterile barrier system delivers consistent results.
Future of Sterile Barrier Systems in Australia
The future of the sterile barrier system in Australia looks promising, with continuous advancements in materials and technology. As regulations evolve, businesses must adapt their sterile barrier system strategies to remain compliant. Innovations in contamination prevention packaging technology and aseptic barrier packaging solutions will drive the next generation of sterile barrier system designs. Companies that embrace these changes will gain a competitive edge in the market.
Conclusion: Investing in Safety and Value
A sterile barrier system is more than just packaging; it is a critical component of healthcare safety and reliability. For Australian businesses, investing in a high-quality sterile barrier system ensures compliance, builds trust, and supports cost-effective operations. By leveraging advanced sterile packaging systems, microbial barrier materials, and contamination prevention packaging technology, companies can achieve optimal results. With trusted partners like vanderstahl, businesses can confidently implement a sterile barrier system that meets both safety standards and budget expectations.